From Wikipedia, the free encyclopedia

Greenhouse effect schematic showing energy flows between space, the atmosphere, and Earth's surface. Energy exchanges are expressed in watts per square meter (W/m2).
Since the beginning of the Industrial Revolution, the burning of fossil fuels has contributed to a 40% increase in the concentration of carbon dioxide in the atmosphere from 280 ppm to 397 ppm, despite the uptake of a large portion of the emissions through various natural "sinks" involved in the carbon cycle.[5][6] Anthropogenic carbon dioxide (CO2) emissions (i.e., emissions produced by human activities) come from combustion of carbon based fuels, principally wood, coal, oil, and natural gas.[7]
[edit] Gases in Earth's atmosphere
[edit] Greenhouse gases
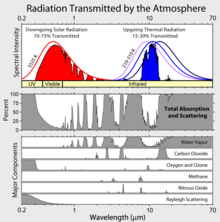
Atmospheric absorption and scattering at different wavelengths of electromagnetic waves. The largest absorption band of carbon dioxide is in the infrared.
- water vapor (H2O)
- carbon dioxide (CO2)
- methane (CH4)
- nitrous oxide (N2O)
- ozone (O3)
[edit] Non-greenhouse gases
Although contributing to many other physical and chemical reactions, the major atmospheric constituents, nitrogen (N2), oxygen (O2), and argon (Ar), are not greenhouse gases. This is because molecules containing two atoms of the same element such as N2 and O2 and monatomic molecules such as Argon (Ar) have no net change in their dipole moment when they vibrate and hence are almost totally unaffected by infrared radiation. Although molecules containing two atoms of different elements such as carbon monoxide (CO) or hydrogen chloride (HCl) absorb IR, these molecules are short-lived in the atmosphere owing to their reactivity and solubility. Because they do not contribute significantly to the greenhouse effect, they are usually omitted when discussing greenhouse gases.[edit] Indirect radiative effects

The false colors in this image represent levels of carbon monoxide in the lower atmosphere, ranging from about 390 parts per billion (dark brown pixels), to 220 parts per billion (red pixels), to 50 parts per billion (blue pixels).[10]
A second type of indirect effect happens when chemical reactions in the atmosphere involving these gases change the concentrations of greenhouse gases. For example, the destruction of non methane volatile organic compounds (NMVOC) in the atmosphere can produce ozone. The size of the indirect effect can depend strongly on where and when the gas is emitted.[11]
Methane has a number of indirect effects in addition to forming CO2. Firstly, the main chemical which destroys methane in the atmosphere is the hydroxyl radical (OH). Methane reacts with OH and so more methane means that the concentration of OH goes down. Effectively, methane increases its own atmospheric lifetime and therefore its overall radiative effect. The second effect is that the oxidation of methane can produce ozone. Thirdly, as well as making CO2 the oxidation of methane produces water; this is a major source of water vapour in the stratosphere which is otherwise very dry. CO and NMVOC also produce CO2 when they are oxidised. They remove OH from the atmosphere and this leads to higher concentrations of methane. The same process that converts them to carbon dioxide can also lead to the formation of tropospheric ozone. Halocarbons have an indirect effect because they destroy stratospheric ozone. Finally hydrogen can lead to ozone production and CH4 increases as well as producing water vapour in the stratosphere.[11]
[edit] Contribution of clouds to Earth's greenhouse effect
The major non-gas contributor to the Earth's greenhouse effect, clouds, also absorb and emit infrared radiation and thus have an effect on radiative properties of the greenhouse gases. Clouds are water droplets or ice crystals suspended in the atmosphere.[12][13][edit] Impact of a given gas on the overall greenhouse effect
The contribution of each gas to the greenhouse effect is affected by the characteristics of that gas, its abundance, and any indirect effects it may cause. For example, on a molecule-for-molecule basis the direct radiative effects of methane is about 72 times stronger than carbon dioxide over a 20 year time frame[14] but it is present in much smaller concentrations so that its total direct radiative effect is smaller, and it has a shorter atmospheric lifetime. On the other hand, in addition to its direct radiative impact methane has a large indirect radiative effect because it contributes to ozone formation. Shindell et al. (2005)[15] argue that the contribution to climate change from methane is at least double previous estimates as a result of this effect.[16]When these gases are ranked by their direct contribution to the greenhouse effect, the most important are:[12]
| Gas | Formula | Contribution (%) |
|---|---|---|
| Water vapor | H2O | 36 – 72% |
| Carbon dioxide | CO2 | 9 – 26% |
| Methane | CH4 | 4 – 9% |
| Ozone | O3 | 3 – 7% |
[edit] Proportion of direct effects at a given moment
It is not possible to state that a certain gas causes an exact percentage of the greenhouse effect. This is because some of the gases absorb and emit radiation at the same frequencies as others, so that the total greenhouse effect is not simply the sum of the influence of each gas. The higher ends of the ranges quoted are for each gas alone; the lower ends account for overlaps with the other gases.[12][13] In addition, some gases such as methane are known to have large indirect effects that are still being quantified.[18][edit] Atmospheric lifetime
Aside from water vapor, which has a residence time of about nine days,[19] major greenhouse gases are well-mixed, and take many years to leave the atmosphere.[20] Although it is not easy to know with precision how long it takes greenhouse gases to leave the atmosphere, there are estimates for the principal greenhouse gases. Jacob (1999)[21] defines the lifetime of an atmospheric species X in a one-box model as the average time that a molecule of X remains in the box. Mathematically
of an atmospheric species X in a one-box model as the average time that a molecule of X remains in the box. Mathematically  can be defined as the ratio of the mass
can be defined as the ratio of the mass 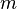 (in kg) of X in the box to its removal rate, which is the sum of the flow of X out of the box (
(in kg) of X in the box to its removal rate, which is the sum of the flow of X out of the box ( ), chemical loss of X (
), chemical loss of X ( ), and deposition of X (
), and deposition of X (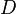 ) (all in kg/sec):
) (all in kg/sec):  .[21] If one stopped pouring any of this gas into the box, then after a time
.[21] If one stopped pouring any of this gas into the box, then after a time  , its concentration would be about halved.
, its concentration would be about halved.The atmospheric lifetime of a species therefore measures the time required to restore equilibrium following a sudden increase or decrease in its concentration in the atmosphere. Individual atoms or molecules may be lost or deposited to sinks such as the soil, the oceans and other waters, or vegetation and other biological systems, reducing the excess to background concentrations. The average time taken to achieve this is the mean lifetime.
Carbon dioxide has a variable atmospheric lifetime, and cannot be specified precisely.[22] The atmospheric lifetime of CO2 is estimated of the order of 30–95 years.[23] This figure accounts for CO2 molecules being removed from the atmosphere by mixing into the ocean, photosynthesis, and a few other processes. However, this excludes the balancing fluxes of CO2 into the atmosphere from the geological reservoirs, which have slower characteristic rates.[24] While more than half of the CO2 emitted is currently removed from the atmosphere within a century, some fraction (about 20%) of emitted CO2 remains in the atmosphere for many thousands of years.[25][26][27]
[edit] Radiative forcing
When energy from the sun reaches the Earth, the planet absorbs some of this energy and radiates the rest back to space as heat.[28] The Earth's surface temperature depends on this balance between incoming and outgoing energy.[28] If this energy balance is shifted, the Earth's surface could become noticeably warmer or cooler, leading to a variety of changes in global climate.[28]A number of natural and man-made mechanisms can affect the global energy balance and force changes in the Earth's climate.[28] Greenhouse gases are one such mechanism.[28] Greenhouse gases in the atmosphere absorb and re-emit some of the outgoing energy radiated from the Earth's surface, causing that heat to be retained in the lower atmosphere.[28] As explained above, some greenhouse gases remain in the atmosphere for decades or even centuries, and therefore can affect the Earth's energy balance over a long time period.[28] Factors that influence Earth's energy balance can be quantified in terms of "radiative climate forcing."[28] Positive radiative forcing indicates warming (for example, by increasing incoming energy or decreasing the amount of energy that escapes to space), while negative forcing is associated with cooling.[28]
[edit] Global warming potential
The global warming potential (GWP) depends on both the efficiency of the molecule as a greenhouse gas and its atmospheric lifetime. GWP is measured relative to the same mass of CO2 and evaluated for a specific timescale. Thus, if a gas has a high (positive) radiative forcing but also a short lifetime, it will have a large GWP on a 20 year scale but a small one on a 100 year scale. Conversely, if a molecule has a longer atmospheric lifetime than CO2 its GWP will increase with the timescale considered. Carbon dioxide is defined to have a GWP of 1 over all time periods.Methane has an atmospheric lifetime of 12 ± 3 years and a GWP of 72 over 20 years, 25 over 100 years and 7.6 over 500 years. The decrease in GWP at longer times is because methane is degraded to water and CO2 through chemical reactions in the atmosphere.
Examples of the atmospheric lifetime and GWP relative to CO2 for several greenhouse gases are given in the following table:[14]
| Gas name | Chemical formula | Lifetime (years) | Global warming potential (GWP) for given time horizon | ||
|---|---|---|---|---|---|
| 20-yr | 100-yr | 500-yr | |||
| Carbon dioxide | CO2 | See above | 1 | 1 | 1 |
| Methane | CH4 | 12 | 72 | 25 | 7.6 |
| Nitrous oxide | N2O | 114 | 289 | 298 | 153 |
| CFC-12 | CCl2F2 | 100 | 11 000 | 10 900 | 5 200 |
| HCFC-22 | CHClF2 | 12 | 5 160 | 1 810 | 549 |
| Tetrafluoromethane | CF4 | 50 000 | 5 210 | 7 390 | 11 200 |
| Hexafluoroethane | C2F6 | 10 000 | 8 630 | 12 200 | 18 200 |
| Sulfur hexafluoride | SF6 | 3 200 | 16 300 | 22 800 | 32 600 |
| Nitrogen trifluoride | NF3 | 740 | 12 300 | 17 200 | 20 700 |
[edit] Natural and anthropogenic sources
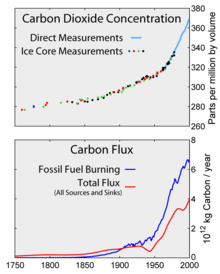
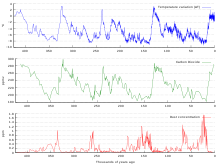
Top: Increasing atmospheric carbon dioxide levels as measured in the atmosphere and reflected in ice cores. Bottom: The amount of net carbon increase in the atmosphere, compared to carbon emissions from burning fossil fuel.
The 2007 Fourth Assessment Report compiled by the IPCC (AR4) noted that "changes in atmospheric concentrations of greenhouse gases and aerosols, land cover and solar radiation alter the energy balance of the climate system", and concluded that "increases in anthropogenic greenhouse gas concentrations is very likely to have caused most of the increases in global average temperatures since the mid-20th century".[33] In AR4, "most of" is defined as more than 50%.
| Gas | Pre-1750 tropospheric concentration[35] | Recent tropospheric concentration[36] | Absolute increase since 1750 | Percentage increase since 1750 | Increased radiative forcing (W/m2)[37] |
|---|---|---|---|---|---|
| Carbon dioxide (CO2) | 280 ppm[38] | 390.5 ppm[39] | 110.5 ppm | 39.5% | 1.79 |
| Methane (CH4) | 700 ppb[40] | 1871 ppb /[41] 1750 ppb[41] | 1171 ppb / 1050 ppb | 167% / 150% | 0.50 |
| Nitrous oxide (N2O) | 270 ppb[37][42] | 323 ppb /[41] 322 ppb[41] | 53 ppb / 52 ppb | 19.6% / 19.3% | 0.18 |
| Tropospheric ozone (O3) | 25 ppb[35] | 34 ppb[35] | 9 ppb | 36% | 0.35[43] |
| Gas | Current (1998) Amount by volume | Radiative forcing (W/m2) |
|---|---|---|
| CFC-11 | 268 ppt | 0.07 |
| CFC-12 | 533 ppt | 0.17 |
| CFC-113 | 84 ppt | 0.03 |
| Carbon tetrachloride | 102 ppt | 0.01 |
| HCFC-22 | 69 ppt | 0.03 |
Ice cores provide evidence for variation in greenhouse gas concentrations over the past 800,000 years (see the following section). Both CO2 and CH4 vary between glacial and interglacial phases, and concentrations of these gases correlate strongly with temperature. Direct data does not exist for periods earlier than those represented in the ice core record, a record that indicates CO2 mole fractions stayed within a range of 180 ppm to 280 ppm throughout the last 800,000 years, until the increase of the last 250 years. However, various proxies and modeling suggests larger variations in past epochs; 500 million years ago CO2 levels were likely 10 times higher than now.[46] Indeed higher CO2 concentrations are thought to have prevailed throughout most of the Phanerozoic eon, with concentrations four to six times current concentrations during the Mesozoic era, and ten to fifteen times current concentrations during the early Palaeozoic era until the middle of the Devonian period, about 400 Ma.[47][48][49] The spread of land plants is thought to have reduced CO2 concentrations during the late Devonian, and plant activities as both sources and sinks of CO2 have since been important in providing stabilising feedbacks.[50] Earlier still, a 200-million year period of intermittent, widespread glaciation extending close to the equator (Snowball Earth) appears to have been ended suddenly, about 550 Ma, by a colossal volcanic outgassing that raised the CO2 concentration of the atmosphere abruptly to 12%, about 350 times modern levels, causing extreme greenhouse conditions and carbonate deposition as limestone at the rate of about 1 mm per day.[51] This episode marked the close of the Precambrian eon, and was succeeded by the generally warmer conditions of the Phanerozoic, during which multicellular animal and plant life evolved. No volcanic carbon dioxide emission of comparable scale has occurred since. In the modern era, emissions to the atmosphere from volcanoes are only about 1% of emissions from human sources.[51][52][53]
[edit] Ice cores
Measurements from Antarctic ice cores show that before industrial emissions started atmospheric CO2 mole fractions were about 280 parts per million (ppm), and stayed between 260 and 280 during the preceding ten thousand years.[54] Carbon dioxide mole fractions in the atmosphere have gone up by approximately 35 percent since the 1900s, rising from 280 parts per million by volume to 387 parts per million in 2009. One study using evidence from stomata of fossilized leaves suggests greater variability, with carbon dioxide mole fractions above 300 ppm during the period seven to ten thousand years ago,[55] though others have argued that these findings more likely reflect calibration or contamination problems rather than actual CO2 variability.[56][57] Because of the way air is trapped in ice (pores in the ice close off slowly to form bubbles deep within the firn) and the time period represented in each ice sample analyzed, these figures represent averages of atmospheric concentrations of up to a few centuries rather than annual or decadal levels.[edit] Changes since the Industrial Revolution
Since the beginning of the Industrial Revolution, the concentrations of most of the greenhouse gases have increased. For example, the mole fraction of carbon dioxide has increased from 280 ppm by about 36% to 380 ppm, or 100 ppm over modern pre-industrial levels. The first 50 ppm increase took place in about 200 years, from the start of the Industrial Revolution to around 1973; however the next 50 ppm increase took place in about 33 years, from 1973 to 2006.[58]Recent data also shows that the concentration is increasing at a higher rate. In the 1960s, the average annual increase was only 37% of what it was in 2000 through 2007.[59]
Today, the stock of carbon in the atmosphere increases by more than 3 million tonnes per annum (0.04%) compared with the existing stock.[clarification needed] This increase is the result of human activities by burning fossil fuels, deforestation and forest degradation in tropical and boreal regions.[60]
The other greenhouse gases produced from human activity show similar increases in both amount and rate of increase. Many observations are available online in a variety of Atmospheric Chemistry Observational Databases.
[edit] Anthropogenic greenhouse gases
This bar graph shows global greenhouse gas emissions by sector from 1990 to 2005, measured in carbon dioxide equivalents.[62]
Modern global anthropogenic carbon emissions.
It is likely that anthropogenic (i.e., human-induced) warming, such as that due to elevated greenhouse gas levels, has had a discernible influence on many physical and biological systems.[66] Future warming is projected to have a range of impacts, including sea level rise,[67] increased frequencies and severities of some extreme weather events,[67] loss of biodiversity,[68] and regional changes in agricultural productivity.[68]
The main sources of greenhouse gases due to human activity are:
- burning of fossil fuels and deforestation leading to higher carbon dioxide concentrations in the air. Land use change (mainly deforestation in the tropics) account for up to one third of total anthropogenic CO2 emissions.[65]
- livestock enteric fermentation and manure management,[69] paddy rice farming, land use and wetland changes, pipeline losses, and covered vented landfill emissions leading to higher methane atmospheric concentrations. Many of the newer style fully vented septic systems that enhance and target the fermentation process also are sources of atmospheric methane.
- use of chlorofluorocarbons (CFCs) in refrigeration systems, and use of CFCs and halons in fire suppression systems and manufacturing processes.
- agricultural activities, including the use of fertilizers, that lead to higher nitrous oxide (N2O) concentrations.
| Seven main fossil fuel combustion sources | Contribution (%) |
|---|---|
| Liquid fuels (e.g., gasoline, fuel oil) | 36% |
| Solid fuels (e.g., coal) | 35% |
| Gaseous fuels (e.g., natural gas) | 20% |
| Cement production | 3 % |
| Flaring gas industrially and at wells | < 1% |
| Non-fuel hydrocarbons | < 1% |
| "International bunker fuels" of transport not included in national inventories | 4 % |
Although CFCs are greenhouse gases, they are regulated by the Montreal Protocol, which was motivated by CFCs' contribution to ozone depletion rather than by their contribution to global warming. Note that ozone depletion has only a minor role in greenhouse warming though the two processes often are confused in the media.
[edit] Role of water vapor
Water vapor accounts for the largest percentage of the greenhouse effect, between 36% and 66% for clear sky conditions and between 66% and 85% when including clouds.[13] Water vapor concentrations fluctuate regionally, but human activity does not significantly affect water vapor concentrations except at local scales, such as near irrigated fields. The atmospheric concentration of vapor is highly variable and depends largely on temperature, from less than 0.01% in extremely cold regions up to 20% in warm, humid regions.[75]The average residence time of a water molecule in the atmosphere is only about nine days, compared to years or centuries for other greenhouse gases such as CH4 and CO2.[76] Thus, water vapor responds to and amplifies effects of the other greenhouse gases. The Clausius-Clapeyron relation establishes that air can hold more water vapor per unit volume when it warms. This and other basic principles indicate that warming associated with increased concentrations of the other greenhouse gases also will increase the concentration of water vapor (assuming that the relative humidity remains approximately constant; modelling and observational studies find that this is indeed so). Because water vapor is a greenhouse gas, this results in further warming and so is a "positive feedback" that amplifies the original warming. Eventually other earth processes offset these positive feedbacks, stabilizing the global temperature at a new equilibrium and preventing the loss of Earth's water through a Venus-like runaway greenhouse effect.[77]
Again assuming constant relative humidity, the Clausius-Clapeyron equation shows that water vapour increases roughly exponentially with temperature, at approximately 7% for typical temperatures.[78]
[edit] Greenhouse gas emissions ("sources")
Between the period 1970 to 2004, GHG emissions (measured in CO2-equivalent)[79] increased at an average rate of 1.6% per year, with CO2 emissions from the use of fossil fuels growing at a rate of 1.9% per year.[80][81] Total anthropogenic emissions at the end of 2009 were estimated at 49.5 gigatonnes CO2-equivalent.[82]:15 These emissions include CO2 from fossil fuel use and from land use, as well as emissions of methane, nitrous oxide and other GHGs covered by the Kyoto Protocol.At present, the two primary sources of CO2 emissions are from burning coal used for electricity generation and petroleum used for motor transport.[citation needed]
[edit] Regional and national attribution of emissions
See also: Kyoto Protocol and government action
There are several different ways of measuring GHG emissions, for example, see World Bank (2010)[83]:362 for tables of national emissions data. Some variables that have been reported[84] include:- Definition of measurement boundaries: Emissions can be attributed geographically, to the area where they were emitted (the territory principle) or by the activity principle to the territory produced the emissions. These two principles result in different totals when measuring, for example, electricity importation from one country to another, or emissions at an international airport.
- Time horizon of different GHGs: Contribution of a given GHG is reported as a CO2 equivalent. The calculation to determine this takes into account how long that gas remains in the atmosphere. This is not always known accurately and calculations must be regularly updated to reflect new information.
- What sectors are included in the calculation (e.g., energy industries, industrial processes, agriculture etc.): There is often a conflict between transparency and availability of data.
- The measurement protocol itself: This may be via direct measurement or estimation. The four main methods are the emission factor-based method, mass balance method, predictive emissions monitoring system, and continuing emissions monitoring systems. These methods differ in accuracy, cost, and usability.
Emissions may be measured over long time periods. This measurement type is called historical or cumulative emissions. Cumulative emissions give some indication of who is responsible for the build-up in the atmospheric concentration of GHGs (IEA, 2007, p. 199).[86]
The national accounts balance would be positively related to carbon emissions. The national accounts balance shows the difference between exports and imports. For many richer nations, such as the United States, the accounts balance is negative because more goods are imported than they are exported. This is mostly due to the fact that it is cheaper to produce goods outside of developed countries, leading the economies of developed countries to become increasingly dependent on services and not goods. We believed that a positive accounts balance would means that more production was occurring in a country, so more factories working would increase carbon emission levels.(Holtz-Eakin, 1995, pp.;85;101).[87]
Emissions may also be measured across shorter time periods. Emissions changes may, for example, be measured against a base year of 1990. 1990 was used in the United Nations Framework Convention on Climate Change (UNFCCC) as the base year for emissions, and is also used in the Kyoto Protocol (some gases are also measured from the year 1995).[71]:146,149 A country's emissions may also be reported as a proportion of global emissions for a particular year.
Another measurement is of per capita emissions. This divides a country's total annual emissions by its mid-year population.[83]:370 Per capita emissions may be based on historical or annual emissions (Banuri et al., 1996, pp. 106–107).[85]
[edit] Greenhouse gas intensity and land-use change
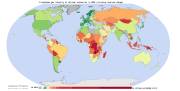
Map of cumulative per capita anthropogenic atmospheric CO2 emissions by country. Cumulative emissions include land use change, and are measured between the years 1950 and 2000.
Land-use change, e.g., the clearing of forests for agricultural use, can affect the concentration of GHGs in the atmosphere by altering how much carbon flows out of the atmosphere into carbon sinks.[89] Accounting for land-use change can be understood as an attempt to measure “net” emissions, i.e., gross emissions from all GHG sources minus the removal of emissions from the atmosphere by carbon sinks (Banuri et al., 1996, pp. 92–93).[85]
There are substantial uncertainties in the measurement of net carbon emissions.[90] Additionally, there is controversy over how carbon sinks should be allocated between different regions and over time (Banuri et al., 1996, p. 93).[85] For instance, concentrating on more recent changes in carbon sinks is likely to favour those regions that have deforested earlier, e.g., Europe.
[edit] Cumulative and historical emissions
Cumulative anthropogenic (i.e., human-emitted) emissions of CO2 from fossil fuel use are a major cause of global warming,[91] and give some indication of which countries have contributed most to human-induced climate change.[92]:15| Region | Industrial CO2 | Total CO2 |
|---|---|---|
| OECD North America | 33.2 | 29.7 |
| OECD Europe | 26.1 | 16.6 |
| Former USSR | 14.1 | 12.5 |
| China | 5.5 | 6.0 |
| Eastern Europe | 5.5 | 4.8 |
Including biotic emissions brings about the same controversy mentioned earlier regarding carbon sinks and land-use change (Banuri et al., 1996, pp. 93–94).[85] The actual calculation of net emissions is very complex, and is affected by how carbon sinks are allocated between regions and the dynamics of the climate system.
Non-OECD countries accounted for 42% of cumulative energy-related CO2 emissions between 1890-2007.[93]:179-180 Over this time period, the US accounted for 28% of emissions; the EU, 23%; Russia, 11%; China, 9%; other OECD countries, 5%; Japan, 4%; India, 3%; and the rest of the world, 18%.[93]:179-180
[edit] Changes since a particular base year
Between 1970-2004, global growth in annual CO2 emissions was driven by North America, Asia, and the Middle East.[94] The sharp acceleration in CO2 emissions since 2000 to more than a 3% increase per year (more than 2 ppm per year) from 1.1% per year during the 1990s is attributable to the lapse of formerly declining trends in carbon intensity of both developing and developed nations. China was responsible for most of global growth in emissions during this period. Localised plummeting emissions associated with the collapse of the Soviet Union have been followed by slow emissions growth in this region due to more efficient energy use, made necessary by the increasing proportion of it that is exported.[70] In comparison, methane has not increased appreciably, and N2O by 0.25% y−1.Using different base years for measuring emissions has an effect on estimates of national contributions to global warming.[92]:17-18[95] This can be calculated by dividing a country's highest contribution to global warming starting from a particular base year, by that country's minimum contribution to global warming starting from a particular base year. Choosing between different base years of 1750, 1900, 1950, and 1990 has a significant effect for most countries.[92]:17-18 Within the G8 group of countries, it is most significant for the UK, France and Germany. These countries have a long history of CO2 emissions (see the section on Cumulative and historical emissions).
[edit] Annual emissions
Energy statistics for fast growing economies are less accurate than those for the industrialized countries. For China's annual emissions in 2008, the Netherlands Environmental Assessment Agency estimated an uncertainty range of about 10%.[96]
[edit] Top emitters
See also: List of countries by carbon dioxide emissions, List of countries by carbon dioxide emissions per capita, List of countries by greenhouse gas emissions, and List of countries by greenhouse gas emissions per capita
[edit] Annual
In 2009, the annual top ten emitting countries accounted for about two-thirds of the world's annual energy-related CO2 emissions.[98]| Country | % of global total annual emissions | Tonnes of GHG per capita |
|---|---|---|
| People's Rep. of China | 23.6 | 5.13 |
| United States | 17.9 | 16.9 |
| India | 5.5 | 1.37 |
| Russian Federation | 5.3 | 10.8 |
| Japan | 3.8 | 8.6 |
| Germany | 2.6 | 9.2 |
| Islamic Rep. of Iran | 1.8 | 7.3 |
| Canada | 1.8 | 15.4 |
| Korea | 1.8 | 10.6 |
| United Kingdom | 1.6 | 7.5 |
[edit] Cumulative
| Country | % of world total | Metric tonnes CO2 per person |
|---|---|---|
| United States | 28.5 | 1,132.7 |
| China | 9.36 | 85.4 |
| Russian Federation | 7.95 | 677.2 |
| Germany | 6.78 | 998.9 |
| United Kingdom | 5.73 | 1,127.8 |
| Japan | 3.88 | 367 |
| France | 2.73 | 514.9 |
| India | 2.52 | 26.7 |
| Canada | 2.17 | 789.2 |
| Ukraine | 2.13 | 556.4 |
[edit] Embedded emissions
One way of attributing greenhouse gas (GHG) emissions is to measure the embedded emissions (also referred to as "embodied emissions") of goods that are being consumed. Emissions are usually measured according to production, rather than consumption.[101] For example, in the main international treaty on climate change (the UNFCCC), countries report on emissions produced within their borders, e.g., the emissions produced from burning fossil fuels.[93]:179[102]:1 Under a production-based accounting of emissions, embedded emissions on imported goods are attributed to the exporting, rather than the importing, country. Under a consumption-based accounting of emissions, embedded emissions on imported goods are attributed to the importing country, rather than the exporting, country.Davis and Caldeira (2010)[102]:4 found that a substantial proportion of CO2 emissions are traded internationally. The net effect of trade was to export emissions from China and other emerging markets to consumers in the US, Japan, and Western Europe. Based on annual emissions data from the year 2004, and on a per-capita consumption basis, the top-5 emitting countries were found to be (in tCO2 per person, per year): Luxembourg (34.7), the US (22.0), Singapore (20.2), Australia (16.7), and Canada (16.6).[102]:5 Carbon Trust research revealed that approximately 25% of all CO2 emissions from human activities 'flow' (i.e. are imported or exported) from one country to another. Major developed economies were found to be typically net importers of embodied carbon emissions - with UK consumption emissions 34% higher than production emissions, and Germany (29%), Japan (19%) and the USA (13%) also significant net importers of embodied emissions.[103]
[edit] Effect of policy
See also: Kyoto Protocol and government action
Governments have taken action to reduce GHG emissions (climate change mitigation). Assessments of policy effectiveness have included work by the Intergovernmental Panel on Climate Change,[104] International Energy Agency,[105][106] and United Nations Environment Programme.[107] Policies implemented by governments have included[108][109][110] national and regional targets to reduce emissions, promoting energy efficiency, and support for renewable energy.Countries and regions listed in Annex I of the United Nations Framework Convention on Climate Change (UNFCCC) (i.e., the OECD and former planned economies of the Soviet Union) are required to submit periodic assessments to the UNFCCC of actions they are taking to address climate change.[110]:3 Analysis by the UNFCCC (2011)[110]:8 suggested that policies and measures undertaken by Annex I Parties may have produced emission savings of 1.5 thousand Tg CO2-eq in the year 2010, with most savings made in the energy sector. The projected emissions saving of 1.5 thousand Tg CO2-eq is measured against a hypothetical "baseline" of Annex I emissions, i.e., projected Annex I emissions in the absence of policies and measures. The total projected Annex I saving of 1.5 thousand CO2-eq does not include emissions savings in seven of the Annex I Parties.[110]:8
[edit] Projections
Further information: climate change scenario#Quantitative emissions projections
A wide range of projections of future GHG emissions have been produced.[111] Rogner et al. (2007)[112] assessed the scientific literature on GHG projections. Rogner et al. (2007)[80] concluded that unless energy policies changed substantially, the world would continue to depend on fossil fuels until 2025-2030. Projections suggest that more than 80% of the world's energy will come from fossil fuels. This conclusion was based on "much evidence" and "high agreement" in the literature.[80] Projected annual energy-related CO2 emissions in 2030 were 40-110% higher than in 2000, with two-thirds of the increase originating in developing countries.[80] Projected annual per capita emissions in developed country regions remained substantially lower (2.8-5.1 tonnes CO2) than those in developed country regions (9.6-15.1 tonnes CO2).[113] Projections consistently showed increase in annual world GHG emissions (the "Kyoto" gases,[114] measured in CO2-equivalent) of 25-90% by 2030, compared to 2000.[80][edit] Relative CO2 emission from various fuels
One liter of gasoline, when used as a fuel, produces 2.32 kg (about 1300 liters or 1.3 cubic meters) of carbon dioxide, a greenhouse gas. One US gallon produces 19.4 lb (1,291.5 gallons or 172.65 cubic feet)[115][116][117]| Fuel name | CO2 emitted (lbs/106 Btu) | CO2 emitted (g/106 J) |
|---|---|---|
| Natural gas | 117 | 50.30 |
| Liquefied petroleum gas | 139 | 59.76 |
| Propane | 139 | 59.76 |
| Aviation gasoline | 153 | 65.78 |
| Automobile gasoline | 156 | 67.07 |
| Kerosene | 159 | 68.36 |
| Fuel oil | 161 | 69.22 |
| Tires/tire derived fuel | 189 | 81.26 |
| Wood and wood waste | 195 | 83.83 |
| Coal (bituminous) | 205 | 88.13 |
| Coal (sub-bituminous) | 213 | 91.57 |
| Coal (lignite) | 215 | 92.43 |
| Petroleum coke | 225 | 96.73 |
| Coal (anthracite) | 227 | 97.59 |
[edit] Removal from the atmosphere ("sinks")
[edit] Natural processes
Greenhouse gases can be removed from the atmosphere by various processes, as a consequence of:- a physical change (condensation and precipitation remove water vapor from the atmosphere).
- a chemical reaction within the atmosphere. For example, methane is oxidized by reaction with naturally occurring hydroxyl radical, OH· and degraded to CO2 and water vapor (CO2 from the oxidation of methane is not included in the methane Global warming potential). Other chemical reactions include solution and solid phase chemistry occurring in atmospheric aerosols.
- a physical exchange between the atmosphere and the other compartments of the planet. An example is the mixing of atmospheric gases into the oceans.
- a chemical change at the interface between the atmosphere and the other compartments of the planet. This is the case for CO2, which is reduced by photosynthesis of plants, and which, after dissolving in the oceans, reacts to form carbonic acid and bicarbonate and carbonate ions (see ocean acidification).
- a photochemical change. Halocarbons are dissociated by UV light releasing Cl· and F· as free radicals in the stratosphere with harmful effects on ozone (halocarbons are generally too stable to disappear by chemical reaction in the atmosphere).
[edit] Negative emissions
See also: Bio-energy with carbon capture and storage, Carbon dioxide air capture, Geoengineering, and Greenhouse gas remediation
A number of technologies remove greenhouse gases emissions from the atmosphere. Most widely analysed are those that remove carbon dioxide from the atmosphere, either to geologic formations such as bio-energy with carbon capture and storage[119][120][121] and carbon dioxide air capture,[121] or to the soil as in the case with biochar.[121] The IPCC has pointed out that many long-term climate scenario models require large scale manmade negative emissions to avoid serious climate change.[122][edit] History of scientific research
Late 19th century scientists experimentally discovered that N2 and O2 do not absorb infrared radiation (called, at that time, "dark radiation") while, on the contrary, water, both as true vapour and condensed in the form of microscopic droplets suspended in clouds, as well as CO2 and other poly-atomic gaseous molecules, do absorb infrared radiation. It was recognized in the early 20th century that greenhouse gases in the atmosphere made the Earth's overall temperature higher than it would be without them. During the late 20th century, a scientific consensus evolved that increasing concentrations of greenhouse gases in the atmosphere are causing a substantial rise in global temperatures and changes to other parts of the climate system, with consequences for the environment and for human health.[123][edit] See also
- Attribution of recent climate change
- Carbon credit
- Carbon Disclosure Project
- Carbon emissions reporting
- Carbon neutrality
- Carbon offset
- Clean Air Act
- Cool Earth 50
- Infrared window Atmospheric window
- Integrated Carbon Observation System
- Eddy covariance (also known as eddy correlation and eddy flux)
- Effects of global warming
- Emission standard
- Environmental impact of aviation
- European Climate Change Programme
- Externality
- Global Atmosphere Watch
- Greenhouse debt
- Hydrogen economy
- List of countries by electricity production from renewable sources
- List of international environmental agreements
- Low-carbon economy
- Low-carbon fuel standard
- Mobile source air pollution
- North American Carbon Program
- Physical properties of greenhouse gases
- Regional Greenhouse Gas Initiative
- Regulation of greenhouse gases under the Clean Air Act
- Sustainability measurement
- World energy consumption
- Zero-emissions vehicle
[edit] Notes
- ^ a b "IPCC AR4 SYR Appendix Glossary" (PDF). http://www.ipcc.ch/pdf/assessment-report/ar4/syr/ar4_syr_appendix.pdf. Retrieved 14 December 2008.
- ^ Karl TR, Trenberth KE (2003). "Modern global climate change". Science 302 (5651): 1719–23. Bibcode 2003Sci...302.1719K. doi:10.1126/science.1090228. PMID 14657489. http://www.sciencemag.org/cgi/content/abstract/302/5651/1719.
- ^ Le Treut H., Somerville R., Cubasch U., Ding Y., Mauritzen C., Mokssit A., Peterson T. and Prather M. (2007) (PDF). Historical overview of climate change science. In: Climate change 2007: The physical science basis. Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change (Solomon S., Qin D., Manning M., Chen Z., Marquis M., Averyt K.B., Tignor M. and Miller H.L., editors). Cambridge University Press. http://www.ipcc.ch/pdf/assessment-report/ar4/wg1/ar4-wg1-chapter1.pdf. Retrieved 14 December 2008.
- ^ "NASA Science Mission Directorate article on the water cycle". Nasascience.nasa.gov. http://nasascience.nasa.gov/earth-science/oceanography/ocean-earth-system/ocean-water-cycle. Retrieved 2010-10-16.
- ^ "Frequently asked global change questions". Carbon Dioxide Information Analysis Center. http://cdiac.ornl.gov/pns/faq.html.
- ^ ESRL Web Team (2008-01-14). "Trends in carbon dioxide". Esrl.noaa.gov. http://www.esrl.noaa.gov/gmd/ccgg/trends/. Retrieved 2011-09-11.
- ^ Lindeburgh, Michael R., (2006). Mechanical engineering reference manual for the PE Exam. Professional Publications, Inc., Belmont, CA. ISBN 978-1-59126-049-3.
- ^ IPCC (2007). "Chapter 7: Couplings Between Changes in the Climate System and Biogeochemistry" (PDF). IPCC WG1 AR4 Report. IPCC. p. FAQ 7.1; report page 512; pdf page 14. http://www.ipcc.ch/pdf/assessment-report/ar4/wg1/ar4-wg1-chapter7.pdf. Retrieved 11 July 2011.
- ^ Canadell, J.G.; Le Quere, C.; Raupach, M.R.; Field, C.B.; Buitenhuis, E.T.; Ciais, P.; Conway, T.J.; Gillett, N.P.; Houghton, R.A.; Marland, G. (2007). "Contributions to accelerating atmospheric CO2 growth from economic activity, carbon intensity, and efficiency of natural sinks". Proc. Natl. Acad. Sci. U.S.A. 104 (47): 18866–18870. Bibcode 2007PNAS..10418866C. doi:10.1073/pnas.0702737104. PMC 2141868. PMID 17962418. //www.ncbi.nlm.nih.gov/pmc/articles/PMC2141868/. Retrieved 15 March 2008.
- ^ http://earthobservatory.nasa.gov/Library/RemoteSensingAtmosphere/remote_sensing6.html[dead link]
- ^ a b Forster, P. et al. (2007). "2.10.3 Indirect GWPs". Changes in Atmospheric Constituents and in Radiative Forcing. Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge University Press. http://www.ipcc.ch/publications_and_data/ar4/wg1/en/ch2s2-10-3.html. Retrieved 2012-12-02.
- ^ a b c Kiehl, J.T.; Kevin E. Trenberth (1997). "Earth's annual global mean energy budget" (PDF). Bulletin of the American Meteorological Society 78 (2): 197–208. Bibcode 1997BAMS...78..197K. doi:10.1175/1520-0477(1997)078<0197:EAGMEB>2.0.CO;2. ISSN 1520-0477. Archived from the original on 30 March 2006. http://web.archive.org/web/20060330013311/http://www.atmo.arizona.edu/students/courselinks/spring04/atmo451b/pdf/RadiationBudget.pdf. Retrieved 1 May 2006.
- ^ a b c "Water vapour: feedback or forcing?". RealClimate. 6 April 2005. http://www.realclimate.org/index.php?p=142. Retrieved 1 May 2006.
- ^ a b IPCC Fourth Assessment Report, Table 2.14, Chap. 2, p. 212
- ^ Shindell, Drew T. (2005). "An emissions-based view of climate forcing by methane and tropospheric ozone". Geophysical Research Letters 32 (4): L04803. Bibcode 2005GeoRL..3204803S. doi:10.1029/2004GL021900. http://www.nasa.gov/vision/earth/lookingatearth/methane.html.
- ^ "Methane's Impacts on Climate Change May Be Twice Previous Estimates". Nasa.gov. 2007-11-30. http://www.nasa.gov/vision/earth/lookingatearth/methane.html. Retrieved 2010-10-16.
- ^ Prather, Michael J.; J Hsu (2008). "NF3, the greenhouse gas missing from Kyoto". Geophysical Research Letters 35 (12): L12810. Bibcode 2008GeoRL..3512810P. doi:10.1029/2008GL034542.
- ^ Isaksen, Ivar S. A.; Michael Gauss, Gunnar Myhre, Katey M. Walter Anthony, and Carolyn Ruppel (20 April 2011). "Strong atmospheric chemistry feedback to climate warming from Arctic methane emissions". Global Biogeochemical Cycles 25 (2). Bibcode 2011GBioC..25B2002I. doi:10.1029/2010GB003845. http://www.atmos.washington.edu/academics/classes/2011Q2/558/IsaksenGB2011.pdf. Retrieved 29 July 2011.
- ^ "AGU Water Vapor in the Climate System". Eso.org. 1995-04-27. http://www.eso.org/gen-fac/pubs/astclim/espas/pwv/mockler.html. Retrieved 2011-09-11.
- ^ Betts (2001). "6.3 Well-mixed Greenhouse Gases". Chapter 6 Radiative Forcing of Climate Change. Working Group I: The Scientific Basis IPCC Third Assessment Report - Climate Change 2001. UNEP/GRID-Arendal - Publications. http://www.grida.no/publications/other/ipcc%5Ftar/?src=/climate/ipcc_tar/wg1/218.htm. Retrieved 2010-10-16.
- ^ a b Jacob, Daniel (1999). Introduction to atmospheric chemistry. Princeton University Press. pp. 25–26. ISBN 0-691-00185-5. http://www-as.harvard.edu/people/faculty/djj/book/.
- ^ "How long will global warming last?". RealClimate. http://www.realclimate.org/index.php/archives/2005/03/how-long-will-global-warming-last. Retrieved 2012-06-12.
- ^ Jacobson, MZ (2005). "Correction to "Control of fossil-fuel particulate black carbon and organic matter, possibly the most effective method of slowing global warming."". J. Geophys. Res. 110: pp. D14105. doi:10.1029/2005JD005888. http://www.agu.org/pubs/crossref/2005/2005JD005888.shtml.
- ^ Archer, David (2009). "Atmospheric lifetime of fossil fuel carbon dioxide". Annual Review of Earth and Planetary Sciences 37: pp. 117–134. doi:10.1146/annurev.earth.031208.100206. http://www.annualreviews.org/doi/abs/10.1146/annurev.earth.031208.100206.
- ^ Meehl, G.A. (2007). "Frequently Asked Question 10.3: If emissions of greenhouse gases are reduced, how quickly do their concentrations in the atmosphere decrease?". In S. Solomon, et al., (eds.). Chapter 10: Global Climate Projections. Climate Change 2007: The Physical Science Basis. Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge University Press (CUP), Cambridge, United Kingdom and New York, NY, USA.: Print version:CUP. This version: IPCC website. http://www.ipcc.ch/publications_and_data/ar4/wg1/en/faq-10-3.html. Retrieved 2011-06-01.
- ^ See also: Archer, David (2005). "Fate of fossil fuel CO2 in geologic time" (PDF). Journal of Geophysical Research 110 (C9): C09S05.1–C09S05.6. Bibcode 2005JGRC..11009S05A. doi:10.1029/2004JC002625. http://geosci.uchicago.edu/~archer/reprints/archer.2005.fate_co2.pdf. Retrieved 27 July 2007.
- ^ See also: Caldeira, Ken; Wickett, Michael E. (2005). "Ocean model predictions of chemistry changes from carbon dioxide emissions to the atmosphere and ocean" (PDF). Journal of Geophysical Research 110 (C9): C09S04.1–12. Bibcode 2005JGRC..11009S04C. doi:10.1029/2004JC002671. Archived from the original on 10 August 2007. http://web.archive.org/web/20070810202611/http://www.ipsl.jussieu.fr/~jomce/acidification/paper/Caldeira_Wickett_2005_JGR.pdf. Retrieved 27 July 2007.
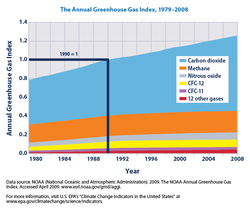
- ^ a b c d e f g h i Edited quote from public-domain source: US Environmental Protection Agency (EPA) (2010). "Climate Change Indicators in the United States". EPA. Greenhouse Gases: Figure 1. The Annual Greenhouse Gas Index, 1979-2008: Background. http://www.epa.gov/climatechange/science/indicators/ghg/climate-forcing.html.. This publication is also available as a PDF (page 18).
- ^ Use of ozone depleting substances in laboratories. TemaNord 2003:516
- ^ Montreal Protocol
- ^ "Chapter 1 Historical Overview of Climate Change Science - FAQ 1.3 Figure 1 description page 116" (PDF). Climate Change 2007: The Physical Science Basis. Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change. Intergovernmental Panel on Climate Change. 5 February 2007. http://www.ipcc.ch/pdf/assessment-report/ar4/wg1/ar4-wg1-chapter1.pdf. Retrieved 25 April 2008.
- ^ "Chapter 3, IPCC Special Report on Emissions Scenarios, 2000". Intergovernmental Panel on Climate Change. 2000. http://www.grida.no/Climate/ipcc/emission/049.htm. Retrieved 2010-10-16.
- ^ "AR4 SYR SPM page 5" (PDF). http://www.ipcc.ch/pdf/assessment-report/ar4/syr/ar4_syr_spm.pdf. Retrieved 2010-10-16.
- ^ From non-copyrighted source: Blasing, T.J. (February 2012), Current Greenhouse Gas Concentrations, doi:10.3334/CDIAC/atg.032, http://cdiac.ornl.gov/pns/current_ghg.html, on CDIAC 2012. Details on copyright status: Frequently Asked Global Change Questions, Q34: I would like to use a diagram, image, graph, table, or other materials from the CDIAC Web site. How can I obtain permission? Are there copyright restrictions?, http://cdiac.ornl.gov/faq.html, retrieved 2012-09-26, on CDIAC 2012. "All of the reports, graphics, data, and other information on the CDIAC Web site are freely and publicly available without copyright restrictions. However as a professional courtesy, we ask that the original data source be acknowledged."
- ^ a b c Ehhalt, D., et al., "Ch 4. Atmospheric Chemistry and Greenhouse Gases", Table 4.1, in IPCC TAR WG1 2001, pp. 244–245. Referred to by: Blasing, T.J. (February 2012), Current Greenhouse Gas Concentrations, doi:10.3334/CDIAC/atg.032, http://cdiac.ornl.gov/pns/current_ghg.html, on CDIAC 2012. Based on Blasing et al. (2012): Inferred global-scale trace-gas concentrations from prior to 1750 are assumed to be practically uninfluenced by human activities such as increasingly specialized agriculture, land clearing, and combustion of fossil fuels. The short atmospheric lifetime of ozone (hours-days) together with the spatial variability of its sources precludes a globally or vertically homogeneous distribution, so that a fractional unit such as parts per billion would not apply over a range of altitudes or geographical locations. Therefore a different unit is used to integrate the varying concentrations of ozone in the vertical dimension over a unit area, and the results can then be averaged globally. This unit is called a Dobson Unit (D.U.), which is the amount of ozone in a column which, unmixed with the rest of the atmosphere, would be 10 micrometers thick at standard temperature and pressure.
- ^ Because atmospheric concentrations of most gases tend to vary systematically over the course of a year, figures given represent averages over a 12-month period for all gases except ozone, for which a current global value has been estimated. CO2 averages for year 2011 are taken from the National Oceanic and Atmospheric Administration, Earth System Research Laboratory, web site: [1] trends maintained by Dr. Pieter Tans. For other chemical species, the values given are averages for 2010. These data are found on the CDIAC AGAGE web site: [2] or the AGAGE home page: [3].
- ^ a b W/m2 = Watts per square metre. Forster, P., et al., "Ch 2: Changes in Atmospheric Constituents and in Radiative Forcing", Table 2.1, in IPCC AR4 WG1 2007, p. 141. Referred to by: Blasing, T.J. (February 2012), Current Greenhouse Gas Concentrations, doi:10.3334/CDIAC/atg.032, http://cdiac.ornl.gov/pns/current_ghg.html, on CDIAC 2012. For the latest updates, see the NOAA Annual Greenhouse Gas Index at: [4].
- ^ ppm = parts-per-million. Prentice, I.C., et al., "Ch 3. The Carbon Cycle and Atmospheric Carbon Dioxide", Executive summary, in IPCC TAR WG1 2001, p. 185. Referred to by: Blasing, T.J. (February 2012), Current Greenhouse Gas Concentrations, doi:10.3334/CDIAC/atg.032, http://cdiac.ornl.gov/pns/current_ghg.html, on CDIAC 2012
- ^ Recent CO2 concentration (390.5 ppm) is the 2011 average taken from globally averaged marine surface data given by the National Oceanic and Atmospheric Administration Earth System Research Laboratory, web site: [5]. Please read the material on that web page and reference Dr. Pieter Tans when citing this average (Dr. Pieter Tans, NOAA/ESRL [6]). The oft-cited Mauna Loa average for 2011 is 391.6, which is a good approximation although 1.1 ppm higher than the spatial averages given above. Refer to [7] for records back to the late 1950s.
- ^ ppb = parts-per-billion
- ^ a b c d The first value in a cell represents Mace Head, Ireland, a mid-latitude Northern-Hemisphere site, and the second value represents Cape Grim, Tasmania, a mid-latitude Southern-Hemisphere site. "Current" values given for these gases are annual arithmetic averages based on monthly background concentrations for year 2010. The SF6 values are from the AGAGE gas chromatography - mass spectrometer (gc-ms) Medusa measuring system. Source: Advanced Global Atmospheric Gases Experiment (AGAGE) data posted on CDIAC web site at: [8]
- ^ The pre-1750 value for N2O is consistent with ice-core records from 10,000 B.C.E. through 1750 C.E.: IPCC, "Summary for policymakers", Figure SPM.1, in IPCC AR4 WG1 2007, p. 3. Referred to by: Blasing, T.J. (February 2012), Current Greenhouse Gas Concentrations, doi:10.3334/CDIAC/atg.032, http://cdiac.ornl.gov/pns/current_ghg.html, on CDIAC 2012
- ^ Changes in stratospheric ozone have resulted in a decrease in radiative forcing of 0.05 W/m2: Forster, P., et al., "Ch 2: Changes in Atmospheric Constituents and in Radiative Forcing", Table 2.12, in IPCC AR4 WG1 2007, p. 204. Referred to by: Blasing, T.J. (February 2012), Current Greenhouse Gas Concentrations, doi:10.3334/CDIAC/atg.032, http://cdiac.ornl.gov/pns/current_ghg.html, on CDIAC 2012
- ^ "Climate Change 2001: The Scientific Basis". Intergovernmental Panel on Climate Change. http://www.grida.no/climate/ipcc_tar/wg1/221.htm. Retrieved 2010-10-16.
- ^ Blasing, TJ (February 2011). "Recent Greenhouse Gas Concentrations". Carbon Dioxide Information Analysis Center. http://cdiac.esd.ornl.gov/pns/current_ghg.html. Retrieved 2011-05-12.
- ^ Image:Phanerozoic Carbon Dioxide.png
- ^ Berner, Robert A. (1994). "GEOCARB II: a revised model of atmospheric CO2 over Phanerozoic time". American Journal of Science 294: 56–91. doi:10.2475/ajs.294.1.56. ISSN 0002-9599. http://www.neotrucks.com/pdf/01.1994.02berner.pdf.
- ^ Royer, DL; RA Berner and DJ Beerling (2001). "Phanerozoic atmospheric CO2 change: evaluating geochemical and paleobiological approaches". Earth-Science Reviews 54 (4): 349–92. Bibcode 2001ESRv...54..349R. doi:10.1016/S0012-8252(00)00042-8.
- ^ Berner, Robert A.; Kothavala, Zavareth (2001). "GEOCARB III: a revised model of atmospheric CO2 over Phanerozoic time". American Journal of Science 301 (2): 182–204. doi:10.2475/ajs.301.2.182. http://www.geology.yale.edu/~ajs/2001/Feb/qn020100182.pdf.
- ^ Beerling, DJ; Berner, RA (2005). "Feedbacks and the co-evolution of plants and atmospheric CO2". Proc. Natl. Acad. Sci. U.S.A. 102 (5): 1302–5. Bibcode 2005PNAS..102.1302B. doi:10.1073/pnas.0408724102. PMC 547859. PMID 15668402. //www.ncbi.nlm.nih.gov/pmc/articles/PMC547859/.
- ^ a b Hoffmann, PF; AJ Kaufman, GP Halverson, DP Schrag (1998). "A neoproterozoic snowball earth". Science 281 (5381): 1342–6. Bibcode 1998Sci...281.1342H. doi:10.1126/science.281.5381.1342. PMID 9721097. http://www.sciencemag.org/cgi/content/full/281/5381/1342.
- ^ Gerlach, TM (1991). "Present-day CO2 emissions from volcanoes". Transactions of the American Geophysical Union 72 (23): 249–55. Bibcode 1991EOSTr..72..249.. doi:10.1029/90EO10192.
- ^ See also: "U.S. Geological Survey". 14 June 2011. http://www.usgs.gov/newsroom/article.asp?ID=2827&from=rss_home#.UHvSclFA92M. Retrieved 15 October 2012.
- ^ Flückiger, Jacqueline (2002). "High-resolution Holocene N2O ice core record and its relationship with CH4 and CO2". Global Biogeochemical Cycles 16: 1010. Bibcode 2002GBioC..16a..10F. doi:10.1029/2001GB001417.
- ^ Friederike Wagner, Bent Aaby and Henk Visscher (2002). "Rapid atmospheric CO2 changes associated with the 8,200-years-B.P. cooling event". Proc. Natl. Acad. Sci. U.S.A. 99 (19): 12011–4. Bibcode 2002PNAS...9912011W. doi:10.1073/pnas.182420699. PMC 129389. PMID 12202744. //www.ncbi.nlm.nih.gov/pmc/articles/PMC129389/.
- ^ Andreas Indermühle, Bernhard Stauffer, Thomas F. Stocker (1999). "Early Holocene Atmospheric CO2 Concentrations". Science 286 (5446): 1815. doi:10.1126/science.286.5446.1815a. "Early Holocene atmospheric CO2 concentrations". Science. http://www.sciencemag.org/cgi/content/full/286/5446/1815a. Retrieved 26 May 2005.
- ^ H.J. Smith, M Wahlen and D. Mastroianni (1997). "The CO2 concentration of air trapped in GISP2 ice from the Last Glacial Maximum-Holocene transition". Geophysical Research Letters 24 (1): 1–4. Bibcode 1997GeoRL..24....1S. doi:10.1029/96GL03700.
- ^ "Monthly Average Carbon Dioxide Concentration, Mauna Loa Observatory" (PDF). Carbon Dioxide Information Analysis Center. 2005. http://cdiac.ornl.gov/trends/co2/graphics/mlo145e_thrudc04.pdf. Retrieved 14 December 2008.
- ^ Dr. Pieter Tans (3 May 2008) "Annual CO2 mole fraction increase (ppm)" for 1959–2007 National Oceanic and Atmospheric Administration Earth System Research Laboratory, Global Monitoring Division (additional details; see also K.A. Masarie, P.P. Tans (1995). "Extension and integration of atmospheric carbon dioxide data into a globally consistent measurement record". J. Geophys. Research 100: 11593–610. Bibcode 1995JGR...10011593M. doi:10.1029/95JD00859.
- ^ Dumitru-Romulus Târziu, Victor-Dan Păcurar (Jan 2011). "Pădurea, climatul și energia" (in (Romanian)). Rev. pădur. 126 (1): 34–39. ISSN 1583-7890. 16720. http://www.revistapadurilor.ro/(16720). Retrieved 2012-06-11.(webpage has a translation button)
- ^ a b US Environmental Protection Agency (EPA) (2010). "Climate Change Indicators in the United States". EPA. Figure 1. The Annual Greenhouse Gas Index, 1979-2008. http://www.epa.gov/climatechange/science/indicators/ghg/climate-forcing.html.
- ^ US Environmental Protection Agency (EPA) (2010). "Climate Change Indicators in the United States". EPA. Figure 2. Global Greenhouse Gas Emissions by Sector, 1990-2005. http://www.epa.gov/climatechange/science/indicators/ghg/global-ghg-emissions.html.
- ^ "Climate Change 2001: Working Group I: The Scientific Basis: figure 6-6". http://www.grida.no/climate/ipcc_tar/wg1/fig6-6.htm. Retrieved 1 May 2006.
- ^ "The present carbon cycle - Climate Change". Grida.no. http://www.grida.no/climate/vital/13.htm. Retrieved 2010-10-16.
- ^ a b IPCC; Solomon, S., D. Qin, M. Manning, Z. Chen, M. Marquis, K.B. Averyt, M. Tignor and H.L. Miller (eds.) (2007). [PDF 7.83 MB "Chapter 7. Couplings Between Changes in the Climate System and Biogeochemistry"]. Climate Change 2007: The Physical Science Basis. Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge, United Kingdom and New York, NY, USA: Cambridge University Press. ISBN 978-0-521-88009-1. PDF 7.83 MB. Retrieved 13 May 2008.
- ^ IPCC (2007d). "6.1 Observed changes in climate and their effects, and their causes". 6 Robust findings, key uncertainties. Climate Change 2007: Synthesis Report. A Contribution of Working Groups I, II, and III to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change (IPCC). Geneva, Switzerland: IPCC. http://www.ipcc.ch/publications_and_data/ar4/syr/en/mains6-1.html.
- ^ a b IPCC (2007d). "6.2 Drivers and projections of future climate changes and their impacts". 6 Robust findings, key uncertainties. Climate Change 2007: Synthesis Report. A Contribution of Working Groups I, II, and III to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change (IPCC). Geneva, Switzerland: IPCC. http://www.ipcc.ch/publications_and_data/ar4/syr/en/mains6-2.html.
- ^ a b IPCC (2007d). "3.3.1 Impacts on systems and sectors". 3 Climate change and its impacts in the near and long term under different scenarios. Climate Change 2007: Synthesis Report. A Contribution of Working Groups I, II, and III to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change (IPCC). Geneva, Switzerland: IPCC. http://www.ipcc.ch/publications_and_data/ar4/syr/en/mains3-3-1.html.
- ^ H. Steinfeld, P. Gerber, T. Wassenaar, V. Castel, M. Rosales, C. de Haan (2006) Livestock’s long shadow. Environmental issues and options. FAO Livestock, Environment and Development (LEAD) Initiative.
- ^ a b Raupach, M.R. et al. (2007). "Global and regional drivers of accelerating CO2 emissions". Proc. Natl. Acad. Sci. U.S.A. 104 (24): 10288–93. Bibcode 2007PNAS..10410288R. doi:10.1073/pnas.0700609104. PMC 1876160. PMID 17519334. http://www.pnas.org/cgi/reprint/0700609104v1.pdf.
- ^ a b c Grubb, M. (July–September 2003). "The economics of the Kyoto protocol" (PDF). World Economics 4 (3). http://www.econ.cam.ac.uk/rstaff/grubb/publications/J36.pdf.
- ^ Lerner & K. Lee Lerner, Brenda Wilmoth (2006). "Environmental issues: essential primary sources". Thomson Gale. http://catalog.loc.gov/cgi-bin/Pwebrecon.cgi?v3=1&DB=local&CMD=010a+2006000857&CNT=10+records+per+page. Retrieved 11 September 2006.
- ^ a b "Kyoto Protocol". United Nations Framework Convention on Climate Change. Home > Kyoto Protocol. http://unfccc.int/kyoto_protocol/items/2830.php.
- ^ a b King, D., et al. (July 2011), "Copenhagen and Cancun", International climate change negotiations: Key lessons and next steps, Oxford, UK: Smith School of Enterprise and the Environment, University of Oxford, p. 12, doi:10.4210/ssee.pbs.2011.0003, http://edition2a.intellimag.com/?id=ssee-july2011 PDF version is also available
- ^ Evans, Kimberly Masters (2005). "The greenhouse effect and climate change". The environment: a revolution in attitudes. Detroit: Thomson Gale. ISBN 0-7876-9082-1.
- ^ "INVENTORY OF U.S. GREENHOUSE GAS EMISSIONS AND SINKS: 1990–2010". U.S. Environmental Protection Agency. 15 April 2012. p. 1.4. http://www.epa.gov/climatechange/Downloads/ghgemissions/US-GHG-Inventory-2012-Main-Text.pdf. Retrieved 2 June 2012.
- ^ Held, I. M.; Soden, B. J. (2000). "Water Vapor Feedback and Global Warming1". Annual Review of Energy and the Environment 25: 441–475. doi:10.1146/annurev.energy.25.1.441.
- ^ S. Rahmstorf, H.J. Schellnhuber: Der Klimawandel. C.H. Beck, 6. Auflage 2007
- ^ Includes the Kyoto "basket" of GHGs
- ^ a b c d e Rogner, H.-H., D. Zhou, R. Bradley. P. Crabbé, O. Edenhofer, B.Hare, L. Kuijpers, M. Yamaguchi (2007). "Executive Summary". In B. Metz, O.R. Davidson, P.R. Bosch, R. Dave, L.A. Meyer. Introduction. Climate Change 2007: Mitigation. Contribution of Working Group III to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge University Press. ISBN 978-0-521-88011-4. http://www.ipcc.ch/publications_and_data/ar4/wg3/en/ch1s1-es.html.
- ^ Rogner, H.-H., D. Zhou, R. Bradley. P. Crabbé, O. Edenhofer, B.Hare, L. Kuijpers, M. Yamaguchi (2007). "1.3.1 Review of the last three decades". In B. Metz, O.R. Davidson, P.R. Bosch, R. Dave, L.A. Meyer. Introduction. Climate Change 2007: Mitigation. Contribution of Working Group III to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge University Press. ISBN 978-0-521-88011-4. http://www.ipcc.ch/publications_and_data/ar4/wg3/en/ch1s1-3.html#1-3-1. This citation clarifies the time period (1970-2004) for the observed emissions trends
- ^ UNEP (November 2011) (PDF), Bridging the Emissions Gap: A UNEP Synthesis Report, Nairobi, Kenya: United Nations Environment Programme (UNEP), ISBN 978-92-807-3229-0, http://www.unep.org/pdf/UNEP_bridging_gap.pdf UNEP Stock Number: DEW/1470/NA
- ^ a b World Bank (2010). "Selected Development Indicators" (PDF). World Development Report 2010: Development and Climate Change. Washington DC, USA: The International Bank for Reconstruction and Development / The World Bank. Tables A1 and A2. doi:10.1596/978-0-8213-7987-5. ISBN 9780821379875. http://siteresources.worldbank.org/INTWDRS/Resources/477365-1327504426766/8389626-1327510418796/Statistical-Annex.pdf.
- ^ a b Bader, N. and Bleichwitz, R. (2009). "Measuring urban greenhouse gas emissions: The challenge of comparability. ''S.A.P.I.EN.S.'' '''2''' (3)". Sapiens.revues.org. http://sapiens.revues.org/index854.html. Retrieved 2011-09-11.
- ^ a b c d e f g h Banuri, T. (1996) (PDF). Equity and social considerations. In: Climate change 1995: Economic and social dimensions of climate change. Contribution of Working Group III to the Second Assessment Report of the Intergovernmental Panel on Climate Change (J.P. Bruce et al. Eds.). This version: Printed by Cambridge University Press, Cambridge, UK, and New York, NY, USA. PDF version: IPCC website. doi:10.2277/0521568544. ISBN 978-0-521-56854-8. http://www.ipcc.ch/ipccreports/sar/wg_III/ipcc_sar_wg_III_full_report.pdf.
- ^ IEA (2007). World energy outlook 2007 edition – China and India insights. International Energy Agency (IEA), Head of Communication and Information Office, 9 rue de la Fédération, 75739 Paris Cedex 15, France. p. 600. ISBN 978-92-64-02730-5. http://www.iea.org/publications/free_new_Desc.asp?PUBS_ID=1927. Retrieved 2010-05-04.
- ^ Holtz-Eakin, D. (1995). "Stoking the fires? CO2 emissions and economic growth". Journal of Public Economics 57 (1): 85–101. doi:10.1016/0047-2727(94)01449-X. http://www.nber.org. Retrieved 2011-04-20.
- ^ Herzog, T. (November 2006). Yamashita, M.B.. ed (PDF). Target: intensity - an analysis of greenhouse gas intensity targets. World Resources Institute. ISBN 1-56973-638-3. http://pdf.wri.org/target_intensity.pdf. Retrieved 2011-04-11.
- ^ IPCC (2007). "Annex I: Glossary J-P". In B. Metz, O.R. Davidson, P.R. Bosch, R. Dave, L.A. Meyer. Climate Change 2007: Mitigation. Contribution of Working Group III to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change. Print version: Cambridge University Press, Cambridge, United Kingdom and New York, NY, USA. This version: IPCC website. ISBN 978-0-521-88011-4. http://www.ipcc.ch/publications_and_data/ar4/wg3/en/annex1sglossary-j-p.html. Retrieved 2011-04-11.
- ^ Markandya, A. (2001). "7.3.5 Cost Implications of Alternative GHG Emission Reduction Options and Carbon Sinks". In B. Metz et al.. Costing Methodologies. Climate Change 2001: Mitigation. Contribution of Working Group III to the Third Assessment Report of the Intergovernmental Panel on Climate Change. Print version: Cambridge University Press, Cambridge, U.K., and New York, N.Y., U.S.A.. This version: GRID-Arendal website. doi:10.2277/0521015022. ISBN 978-0-521-01502-8. http://www.grida.no/climate/ipcc_tar/wg3/293.htm. Retrieved 2011-04-11.
- ^ Botzen, W.J.W., et al. (2008). [dead link] "Cumulative CO2 emissions: shifting international responsibilities for climate debt". Climate Policy (Earthscan) 8: 570. doi:10.3763/cpol.2008.0539. ISSN (print), 1752-7457 (online) 1469-3062 (print), 1752-7457 (online). http://globalcitizen.net/Data/Pages/1291/papers/2009103014156814.pdf[dead link].
- ^ a b c Höhne, N., et al. (24 September 2010). "Contributions of individual countries’ emissions to climate change and their uncertainty" (PDF). Climatic Change (Springer Science+Business Media B.V.). doi:10.1007/s10584-010-9930-6. http://www.gcca.eu/usr/documents/Contributions_Individual_countries_201011229410.pdf.
- ^ a b c International Energy Agency (IEA) (2009) (PDF), World Energy Outlook 2009, Paris, France: IEA, pp. 179–180, ISBN 978-92-64-06130-9, http://www.iea.org/textbase/nppdf/free/2009/weo2009.pdf
- ^ Rogner, H.-H., D. Zhou, R. Bradley. P. Crabbé, O. Edenhofer, B.Hare, L. Kuijpers, M. Yamaguchi (2007), "1.3.1 Review of the last three decades", in B. Metz, O.R. Davidson, P.R. Bosch, R. Dave, L.A. Meyer, Introduction, Climate Change 2007: Mitigation. Contribution of Working Group III to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change, Cambridge University Press, ISBN 978-0-521-88011-4, http://www.ipcc.ch/publications_and_data/ar4/wg3/en/ch1s1-3.html#1-3-1
- ^ The cited paper uses the term "start date" instead of "base year."
- ^ a b c PBL (25 June 2009). "Global CO2 emissions: annual increase halves in 2008". Netherlands Environmental Assessment Agency (PBL) website. http://www.pbl.nl/en/publications/2009/Global-CO2-emissions-annual-increase-halves-in-2008.html. Retrieved 2010-05-05.
- ^ Carbon Trust (March 2009). "Global Carbon Mechanisms: Emerging lessons and implications (CTC748)". Carbon Trust website. p. 24. http://www.carbontrust.com/resources/reports/advice/global-carbon-mechanisms. Retrieved 2010-03-31.
- ^ International Energy Agency (IEA) (2011), CO2 Emissions From Fuel Combustion: Highlights (2011 edition), Paris, France: IEA, p. 9, http://www.iea.org/publications/free_new_Desc.asp?PUBS_ID=2450
- ^ International Energy Agency (IEA) (2011) (XLS), CO2 Emissions From Fuel Combustion: Highlights (2011 edition): Excel spreadsheet, Paris, France: IEA, http://www.iea.org/co2highlights/CO2highlights.xls
- ^ World Resources Institute (WRI) (2011), Climate Analysis Indicators Tool (CAIT): Indicators: GHG Emissions: Cumulative Emissions (free registration required), Washington DC, USA: WRI, http://cait.wri.org/cait.php
- ^ Helm, D., et al. (10 December 2007) (PDF). Too Good To Be True? The UK's Climate Change Record. Website of Dieter Helm, a professor at the University of Oxford and a Fellow of New College, Oxford. p. 3. http://www.dieterhelm.co.uk/sites/default/files/Carbon_record_2007_0.pdf.
- ^ a b c Davis, S.K. and K. Caldeira (8 March 2010). "Consumption-based Accounting of CO2 Emissions" (PDF). Proceedings of the National Academy of Sciences Early Edition 107 (12). Bibcode 2010PNAS..107.5687D. doi:10.1073/pnas.0906974107. http://www.pnas.org/content/early/2010/02/23/0906974107.full.pdf+html. Retrieved 2011-04-18.
- ^ "International Carbon Flows". Carbon Trust. May 2011. http://www.carbontrust.com/resources/reports/advice/international-carbon-flows. Retrieved 12 Nov 2012.
- ^ e.g., Gupta et al. (2007) assessed the scientific literature on climate change mitigation policy: Gupta, S., et al. (2007). Chapter 13: Policies, instruments, and co-operative arrangements. In: Climate Change 2007: Mitigation. Contribution of Working Group III to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change (B. Metz, O.R. Davidson, P.R. Bosch, R. Dave, L.A. Meyer (eds)). Cambridge University Press. ISBN 9780521880114. http://www.ipcc.ch/publications_and_data/ar4/wg3/en/ch13.html.
- ^ International Energy Agency (IEA) (2012). "International Energy Agency - Energy Policy". Paris, France: IEA. http://iea.org/subjectqueries/keyresult.asp?KEYWORD_ID=4151.
- ^ Organization for Economic Co-operation and Development (OECD) / International Energy Agency (IEA) (2012). "IEA Publications Bookshop: IEA Publications on 'Energy Policy'". Paris, France: OECD/IEA. http://iea.org/w/bookshop/b.aspx?Subject=Energy%20Policy.
- ^ United Nations Environment Programme (UNEP) (November 2011) (PDF), Bridging the Emissions Gap: A UNEP Synthesis Report, Nairobi, Kenya: UNEP, ISBN 978-92-807-3229-0, http://www.unep.org/pdf/UNEP_bridging_gap.pdf UNEP Stock Number: DEW/1470/NA
- ^ World Bank (2010). "4. Energizing development without compromising the climate" (PDF). World Development Report 2010: Development and Climate Change. Washington DC, USA: The International Bank for Reconstruction and Development / The World Bank. p. 192. doi:10.1596/978-0-8213-7987-5. ISBN 9780821379875. http://siteresources.worldbank.org/INTWDRS/Resources/477365-1327504426766/8389626-1327510418796/Chapter-4.pdf.
- ^ United Nations Framework Convention on Climate Change (UNFCCC) (2005) (PDF). Sixth compilation and synthesis of initial national communications from Parties not included in Annex I to the Convention. Note by the secretariat. Executive summary.. Geneva, Switzerland: United Nations Office. pp. 10–12. http://unfccc.int/resource/docs/2005/sbi/eng/18.pdf.
- ^ a b c d United Nations Framework Convention on Climate Change (UNFCCC) (2011) (PDF). Compilation and synthesis of fifth national communications. Executive summary. Note by the secretariat.. Geneva (Switzerland): United Nations Office at Geneva. pp. 9–10. http://unfccc.int/resource/docs/2011/sbi/eng/inf01.pdf.
- ^ Fisher, B., et al. (2007). "3.1 Emissions scenarios". In B. Metz, O.R. Davidson, P.R. Bosch, R. Dave, L.A. Meyer. Chapter 3: Issues related to mitigation in the long-term context. Climate Change 2007: Mitigation. Contribution of Working Group III to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge University Press. ISBN 978-0-521-88011-4. http://www.ipcc.ch/publications_and_data/ar4/wg3/en/ch3s3-1.html.
- ^ Rogner, H.-H., D. Zhou, R. Bradley. P. Crabbé, O. Edenhofer, B.Hare, L. Kuijpers, M. Yamaguchi (2007). "1.3.2 Future outlook". In B. Metz, O.R. Davidson, P.R. Bosch, R. Dave, L.A. Meyer. Introduction. Climate Change 2007: Mitigation. Contribution of Working Group III to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge University Press. ISBN 978-0-521-88011-4. http://www.ipcc.ch/publications_and_data/ar4/wg3/en/ch1s1-3-2.html.
- ^ Rogner, H.-H., D. Zhou, R. Bradley. P. Crabbé, O. Edenhofer, B.Hare, L. Kuijpers, M. Yamaguchi (2007). "1.3.2.4 Total GHG emissions". In B. Metz, O.R. Davidson, P.R. Bosch, R. Dave, L.A. Meyer. Introduction. Climate Change 2007: Mitigation. Contribution of Working Group III to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge University Press. ISBN 978-0-521-88011-4. http://www.ipcc.ch/publications_and_data/ar4/wg3/en/ch1s1-3-2-4.html.
- ^ carbon dioxide, methane, nitrous oxide, sulfur hexafluoride
- ^ "Greenhouse Gas Emissions from a Typical Passenger Vehicle, US Environment Protection Agency". Epa.gov. http://www.epa.gov/otaq/climate/documents/420f11041.pdf. Retrieved 2011-09-11.
- ^ Engber, Daniel (2006-11-01). "How gasoline becomes CO2, Slate Magazine". Slate.com. http://www.slate.com/id/2152685/. Retrieved 2011-09-11.
- ^ "Volume calculation for carbon dioxide". Icbe.com. http://www.icbe.com/carbondatabase/CO2volumecalculation.asp. Retrieved 2011-09-11.
- ^ "Voluntary Reporting of Greenhouse Gases Program". Energy Information Administration. http://www.eia.doe.gov/oiaf/1605/coefficients.html. Retrieved 21 August 2009.
- ^ Obersteiner M, Azar C, Kauppi P et al. (October 2001). "Managing climate risk". Science 294 (5543): 786–7. doi:10.1126/science.294.5543.786b. PMID 11681318.
- ^ Azar, C., Lindgren, K., Larson, E.D. and Möllersten, K. (2006). "Carbon capture and storage from fossil fuels and biomass – Costs and potential role in stabilising the atmosphere". Climatic Change 74: 47–79. doi:10.1007/s10584-005-3484-7. http://www.environmental-expert.com/Files%5C6063%5Carticles%5C6220%5Cw30h4274h130580u.pdf.
- ^ a b c "Geoengineering the climate: science, governance and uncertainty". The Royal Society. 2009. http://royalsociety.org/displaypagedoc.asp?id=35151. Retrieved 12 September 2009.[dead link]
- ^ Fischer, B.S., N. Nakicenovic, K. Alfsen, J. Corfee Morlot, F. de la Chesnaye, J.-Ch. Hourcade, K. Jiang, M. Kainuma, E. La Rovere, A. Matysek, A. Rana, K. Riahi, R. Richels, S. Rose, D. van Vuuren, R. Warren, (2007)“Issues related to mitigation in the long term context”, In Climate Change 2007: Mitigation. Contribution of Working Group III to the Fourth Assessment Report of the Inter-governmental Panel on Climate Change [B. Metz, O.R. Davidson, P.R. Bosch, R. Dave, L.A. Meyer (eds)], Cambridge University Press, Cambridge.
- ^ "Global Warming Opposing Viewpoints Paper". OPPapers. http://www.oppapers.com/essays/Global-Warming-Opposing-Viewpoints-Paper/733173?topic. Retrieved 12 September 2011.
[edit] References
- CDIAC (2012), Carbon Dioxide Information Analysis Center (CDIAC), Oak Ridge, Tenessee, USA: CDIAC, http://cdiac.ornl.gov/
- IPCC TAR WG1 (2001), Houghton, J.T.; Ding, Y.; Griggs, D.J.; Noguer, M.; van der Linden, P.J.; Dai, X.; Maskell, K.; and Johnson, C.A., ed., Climate Change 2001: The Scientific Basis, Contribution of Working Group I (WG1) to the Third Assessment Report (TAR) of the Intergovernmental Panel on Climate Change (IPCC), Cambridge University Press, ISBN 0-521-80767-0, http://www.grida.no/publications/other/ipcc%5Ftar/?src=/climate/ipcc_tar/wg1/index.htm (pb: 0-521-01495-6)
- IPCC AR4 WG1 (2007), Solomon, S.; Qin, D.; Manning, M.; Chen, Z.; Marquis, M.; Averyt, K.B.; Tignor, M.; and Miller, H.L., ed., Climate Change 2007: The Physical Science Basis, Contribution of Working Group I (WG1) to the Fourth Assessment Report (AR4) of the Intergovernmental Panel on Climate Change (IPCC), Cambridge University Press, ISBN 978-0-521-88009-1, http://www.ipcc.ch/publications_and_data/ar4/wg1/en/contents.html (pb: 978-0-521-70596-7)
- VAN DIJK, P., ZHANG, J., JUN, W., KUENZER, C. and K.H. WOLF, 2011: Assessment of the contribution of in-situ combustion of coal to greenhouse gas emission; based on a comparison of Chinese mining information to previous remote sensing estimates. International Journal of Coal Geology, Special Issue RS/GIS, DOI: 10.1016/j.coal.2011.01.009; pp. 108–119
[edit] External links
| Wikisource has original text related to this article: |
- Greenhouse gas at the Open Directory Project
- Annual Greenhouse Gas Index (AGGI) from NOAA
- Atmospheric spectra of GHGs and other trace gases
- How Much Greenhouse Gas Does the United States Emit?
- Grist article on convenient summary from various sources incl IPCC of greenhouse gas emissions * *
- Convenient summary of Greenhouse gas emissions
- Greenhouse Gases
- Greenhouse Gases Sources, Levels, Study results — University of Michigan; eia.doe.gov findings
- EM-1 | Greenhouse gas emissions research project
- Carbon dioxide emissions
- Carbon Emissions World Map in 2009 Mark McCormick and Paul Scruton, The Guardian February 2011
- International Energy Annual: Reserves
- International Energy Annual 2003: Carbon Dioxide Emissions
- International Energy Annual 2003: Notes and Sources for Table H.1co2 (Metric tons of carbon dioxide can be converted to metric tons of carbon equivalent by multiplying by 12/44)
- Textbook on Eddy Covariance Measurements of Gas Emissions
- Trends in Atmospheric Carbon Dioxide (NOAA)
- NOAA Paleoclimatology Program — Vostok Ice Core
- NOAA CMDL CCGG — Interactive Atmospheric Data Visualization NOAA CO2 data
- Carbon Dioxide Information Analysis Centre FAQ Includes links to Carbon Dioxide statistics
- Little Green Data Book 2007, World Bank. Lists CO2 statistics by country, including per capita and by country income class.
- Database of carbon emissions of power plants
- NASA's Orbiting Carbon Observatory
- The Carbon Bag: the carbon dioxide emission of a typical British home
- Methane emissions
- BBC News — Thawing Siberian bogs are releasing more methane
- Textbook on Eddy Covariance Measurements of Gas Emissions












Hey, there is a broken link in this article, under the anchor text - Database of carbon emissions of power plants
ReplyDeleteHere is the working link so you can replace it - https://www.selectra.co.uk/energy/guides/market/carbon-monitoring-for-action